Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...

Watanabe, Masashi; Seki, Takayuki*
Materials Science & Engineering B, 272, p.115369_1 - 115369_6, 2021/10
Times Cited Count:1 Percentile:4.66(Materials Science, Multidisciplinary)The effect of oxygen non-stoichiometry on the initial sintering behavior of CeO was investigated. It was found that the initial sintering of the stoichiometric and hypo-stoichiometric composition was controlled by the grain boundary diffusion. The activation energies of cation diffusion were derived from initial sintering data. Moreover, it is suggested that the cation diffusion was caused by a vacancy mechanism.
was investigated. It was found that the initial sintering of the stoichiometric and hypo-stoichiometric composition was controlled by the grain boundary diffusion. The activation energies of cation diffusion were derived from initial sintering data. Moreover, it is suggested that the cation diffusion was caused by a vacancy mechanism.

Watanabe, Masashi; Matsumoto, Taku; Hirooka, Shun; Morimoto, Kyoichi; Kato, Masato
2018 GIF Symposium Proceedings (Internet), p.315 - 320, 2020/05
Recently, a research group studying at Plutonium Fuel Development Facility (PFDF) in Japan Atomic Energy Agency has systematically measured vast amounts of physical properties in the non-stoichiometric (U, Pu)O . Lattice parameter, elastic modulus, thermal expansion, oxygen potential, oxygen chemical diffusion coefficient and thermal conductivity were successfully measured as function of Pu content, O/M ratio and temperature, and the effects of Pu content and O/M ratio on their physical properties were evaluated. In this work, those experimental data are reviewed, and latest experimental data set on the non-stoichiometric (U, Pu)O
. Lattice parameter, elastic modulus, thermal expansion, oxygen potential, oxygen chemical diffusion coefficient and thermal conductivity were successfully measured as function of Pu content, O/M ratio and temperature, and the effects of Pu content and O/M ratio on their physical properties were evaluated. In this work, those experimental data are reviewed, and latest experimental data set on the non-stoichiometric (U, Pu)O are presented. The data set would be available in development of a fuel performance code.
are presented. The data set would be available in development of a fuel performance code.

Watanabe, Masashi; Sunaoshi, Takeo*; Kato, Masato
Defect and Diffusion Forum, 375, p.84 - 90, 2017/05
The oxygen chemical diffusion coefficient in (U, Pu)O was determined by thermo-gravimetry as functions of the Pu content, oxygen-to-metal ratio and temperature. The surface reaction was considered in the diffusion coefficient determination. The activation energy for the chemical diffusion coefficient was 60 kJ/mol and 65 kJ/mol, respectively, in (U
was determined by thermo-gravimetry as functions of the Pu content, oxygen-to-metal ratio and temperature. The surface reaction was considered in the diffusion coefficient determination. The activation energy for the chemical diffusion coefficient was 60 kJ/mol and 65 kJ/mol, respectively, in (U Pu
Pu )O
)O and (U
and (U Pu
Pu )O
)O .
.
 TiO
TiO
Hoshino, Tsuyoshi; Kobayashi, Takeshi*; Nashimoto, Makoto*; Kawamura, Hiroshi; Terai, Takayuki*; Yamawaki, Michio*; Takahashi, Yoichi*
Journal of the Ceramic Society of Japan, Supplement, Vol.112, No.1 (CD-ROM), p.S354 - S357, 2004/05
no abstracts in English
Yamawaki, Michio*; Yamaguchi, Kenji; Suzuki, Atsushi*
Ionics, 7(4-6), p.339 - 345, 2001/07
Times Cited Count:6 Percentile:36.46(Chemistry, Physical)no abstracts in English
 -PrO
-PrO

 -O
-O system
system; Fujino, Takeo; Tagawa, Hiroaki
Journal of Nuclear Materials, 132, p.192 - 201, 1985/00
Times Cited Count:21 Percentile:89.24(Materials Science, Multidisciplinary)no abstracts in English
 O
O
Fujino, Takeo; ;
Journal of Nuclear Materials, 97, p.93 - 103, 1981/00
Times Cited Count:18 Percentile:87.04(Materials Science, Multidisciplinary)no abstracts in English
; Fujino, Takeo
Inorg.Nucl.Chem.Lett., 16(2), p.91 - 96, 1980/00
no abstracts in English
Fujino, Takeo; Tagawa, Hiroaki; ;
Analytica Chimica Acta, 98(2), p.373 - 383, 1978/02
Times Cited Count:3no abstracts in English
 U
U N
N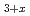
Masaki, N.; Tagawa, Hiroaki
Journal of Nuclear Materials, 57(2), p.187 - 192, 1975/01
Times Cited Count:13no abstracts in English
 Ce
Ce )O
)O solid solution
solid solutionWatanabe, Masashi; Kato, Masato; Sunaoshi, Takeo*; Yato, Tadao*; Konashi, Kenji*
no journal, ,
Raman spectroscopy has been widely used to detect the oxygen vacancies and proven to be a very powerful characterization technique. The Raman spectral data of non-stoichiometric actinide oxides are very limited. The purpose of this work was to measure the Raman spectra of a non-stoichiometric (U Ce
Ce )O
)O solid solution. The Raman spectra of stoichiometric and non-stoichiometric (U
solid solution. The Raman spectra of stoichiometric and non-stoichiometric (U Ce
Ce )O
)O are successfully measured. The T
are successfully measured. The T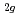 peak of non-stoichiometric (U
peak of non-stoichiometric (U Ce
Ce )O
)O exhibited the same peak shift behavior as hypo-stoichiometric CeO
exhibited the same peak shift behavior as hypo-stoichiometric CeO .
.